Feeding is an innate behavior in the animal kingdom. Proper amount of food consumption is critical for survival and health, as well as for producing high-quality progenies. Although appetite signals for promoting eating have been widely investigated, the mechanisms underlying feeding termination are poorly understood. On Jan 19th, 2017, a research paper entitled “Drosophila FIT Is a Protein-specific Satiety Hormone Essential for Feeding Control” in the journal of Nature Communication addressed this important question.
Animals consume food containing essential amino acids daily, and deficiency in protein consumption leads to severe developmental defects. On the other hand, a bunch of evidences have shown that excess protein intake results in stresses in organs, like kidney and liver, the acid-base imbalance, and even severe diseases, such as renal failure. Among the three macronutrients, protein exerts the greatest inhibitory effect in feeding control. Nevertheless, protein-specific nutrient sensing signals have not been identified, thus, how internal protein hemostasis and protein feeding are regulated await elucidation.
Professor LI Yan and her colleagues at the Institute of Biophysics, Chinese Academy of Sciences utilized Drosophila as the animal model and designed a pre-feeding paradigm to uncover the novel molecules underlying protein feeding. They identified a gene called Fit (Female-specific Independent of Transformer), which expression is dramatically elevated following pre-feeding of protein food, but not other types of food. Overexpression of FIT suppresses protein feeding; on the contrary, Fit mutant flies fail to stop eating after a big meal of protein.
With bioinformatics analysis and experimental evidences, the researchers revealed that FIT is a fat body-secreted peptide, circling in the fly haemolymph. Furthermore, secreted FIT promotes the release of Drosophila insulin-like peptide 2 (DIPL2) in the brain, thereby regulating feeding behavior through the insulin signaling. Interestingly, Fit is a sexually dimorphic gene, with about 10 fold higher levels in females than that in males. In consistent, protein intake-induced DILP2 release and feeding inhibition are also much more evident in female flies, while little in males.
This study illustrated how a single type of nutrients is sensed and conveyed to the central nervous system for a precise regulation of feeding behavior, and thus triggered wide interests. One of the reviewers pointed out that “…Surprisingly, however, the mechanisms by which dietary amino acids induce satiety are not known. The present paper, thus, is a very valuable effort to clarify a signal pathway whereby amino acids regulate satiety and food intake.” Professor LI has been invited to present this work in several first-class international meetings, including the EMBO Conference Series on “the Molecular and Developmental Biology of Drosophila”, and the Gordon Research Conferences on “Modulation of Neural Circuits & Behavior” and “Molecular and Cellular Neurobiology”. She was also invited by Professor Greg S.B. Suh to give a seminar in the Skirball Institute at the New York University, School of Medicine.
Obesity has become a severe health problem in the human society. Identification of the satiety signal sheds lights on the molecular and neural basis for feeding termination, as well as the biotherapy of diet control.
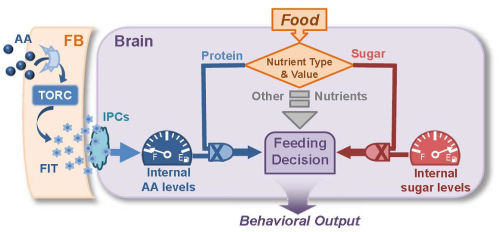
FIT conveys the signal of internal AA levels from the Fat Body (FB) to the brain for nutrient sensing and feeding control (Image by IBP)
This work was supported by grants from the National Science Foundation of China.
Contact:
LI Yan
Institute of Biophysics
Email: liyan@sun5.ibp.ac.cn
Tel: 86-10-64888533
Fax: 86-10-64888533