As the biggest mucosal layer of the host, intestine provides a pivotal interface between the symbiotic/pathogenic microorganisms and the host. As the first line of immune defense, the innate immune response therefore is critical to eliminate invading pathogens, tolerate commensal microbiota and restore the homeostasis of intestinal epithelia. However, excessive inflammation in the gut leads to dysfunction of the epithelial barrier and is causative of inflammatory bowel disease (locally) and even asthma and type I diabetes (systemically). Hence, a better understanding of the innate immune homeostasis in the intestines is increasingly important and necessary.
To identify novel regulators of intestinal innate immune signaling, couple pilot genetic screenings were performed using Drosophila melanogaster (fruit fly), a widely used model organism for their mucosal structure and immune signaling are conserved in mammals.
Professor PAN Lei of the Institute of Biophysics (IBP) of the Chinese Academy of Sciences in cooperation with Professor TANG Hong (Institute Pasteur of Shanghai) and colleagues found that flies deficient of intestinal Bap180 (Baf180 in mammals), a subunit of chromatin remodeling SWI/SNF complex, succumb to excessive inflammation rather than bacterial overload after bacterial infection. Bap180 is induced by Drosophila intestinal innate immune pathway, IMD–Relish signalling (TNFR/IL-1R in mammals), response to bacterial challenge. Thus,upregulated Bap180 functions as a negative feedback regulator to limit overreactive IMD signaling through interfering with Relish binding activity as well as to avoid overexpression of pro-inflammatory gene eiger (TNF in mammals), a critical step to prevent of excessive tissue damage and elongate life span of flies, at transcriptional level. Furthermore, intestinal targeting of Baf180 renders mice susceptible to a more aggressive infectious colitis caused by Citrobacter rodentium. Last, they found that Bap180 not only plays an essential role in preventing the intestinal hyper-inflammatory response to extrinsic pathogen infections, but also maintains the homeostasis of intestinal inflammation intrinsically, in present of commensal bacteria.
Together, their research identified a novel conserved transcriptional repressor, Bap180/Baf180, can feed back to maintain homeostasis of intestinal innate immunity. This study provides a new insight into the transcriptional control of immune-inflammatory response, and may pave the way to discover a potential biomarker or target for treatment of intestinal inflammatory diseases.
The research work, entitled Bap180/Baf180 is required to maintain homeostasis of intestinal innate immune response in Drosophila and mice, was published online in the journal Nature Microbiology on April 18, 2017. This work was supported by grants from the National Natural Science Foundation of China, CAS Youth Innovation Promotion Association and the Ministry of Science and Technology 973 Project.
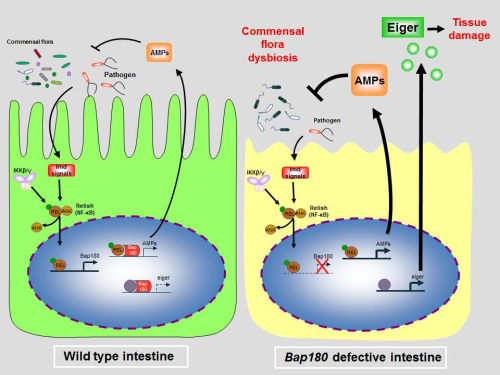
Fig. Bap180 contributes to intestinal innate immune homeostasis in Drosophila (Image by IBP)
Contact:
PAN Lei
CAS Key Laboratory of Infection and Immunity
Institute of Biophysics, Chinese Academy of Sciences
86-10-64881169
panlei@moon.ibp.ac.cn