Long noncoding RNAs (lncRNAs) have emerged as important regulators in almost all biological processes, ranging from maintenance of embryonic stem cell pluripotency, iPS reprogramming, cell cycle regulation to cell differentiation and development. The previously regarded “dark matter” of the human genome has revealed its fundamental roles in life. During the last decade, the exploration of how lncRNAs regulate gene expression has drawn dramatic attention worldwide.
Adipogenesis is a well-orchestrated multistep process of cell differentiation by which preadipocytes become mature adipocytes, which requires the sequential activation of numerous transcription factors, including the CCAAT/enhancer-binding protein (C/EBP) gene family and peroxisome proliferator activated receptor-γ (PPAR-γ). Dysregulation of adipogenesis might lead to metabolic diseases such as obesity. C/EBPα is one of the essential transcription factors. However, the molecular mechanisms by which the expression of C/EBPα is activated are still poorly understood.
In a recent study, XIAO Tengfei and colleagues revealed that long noncoding RNA ADINR regulates the expression of C/EBPα by recruiting chromatin modification complex MLL3/4 to alter the H3K4me3 modification level of the C/EBPα promoter. The study is entitled “Long Noncoding RNA ADINR Regulates Adipogenesis by Transcriptionally Activating C/EBPα” and published online on Stem Cell Reports, led by Prof. CHEN Runsheng from Institute of Biophysics (IBP) of Chinese Academy of Sciences and Prof. Robert Chunhua Zhao from Institute of Basic Medical Sciences , Chinese Academy of Medical Sciences.
Using a combined lncRNA-mRNA microarray designed by Prof. CHEN’s group, researchers identified differentially expressed lncRNAs during induced adipogenesis of cultured human mesenchymal stem cells. LncRNA ADINR and the protein coding gene C/EBPα form a divergent transcription pair in the genomic locus and co-upregulated during adipogenic differentiation. Disruption of ADINR’s expression inhibits the transcription of C/EBPα and perturbs adipogenesis in vitro and in vivo.
The study showed that lncRNA ADINR acts in cis to recruit chromatin modification complex MLL3/4 to the promoter region of C/EBPα by direct binding to one of the MLL3/4 components PA1. Through interacting with ADINR, MLL3/4 complex locates to the promoter of C/EBPα, catalyzing H3K4me3, which facilitates the activation of C/EBPα transcription.
This study revealed the precise mechanism of how divergent lncRNA ADINR regulates its neighboring gene, and it is for the first time to illustrate how lncRNA participates in the genetic control of adipogenic differentiation. Full Text
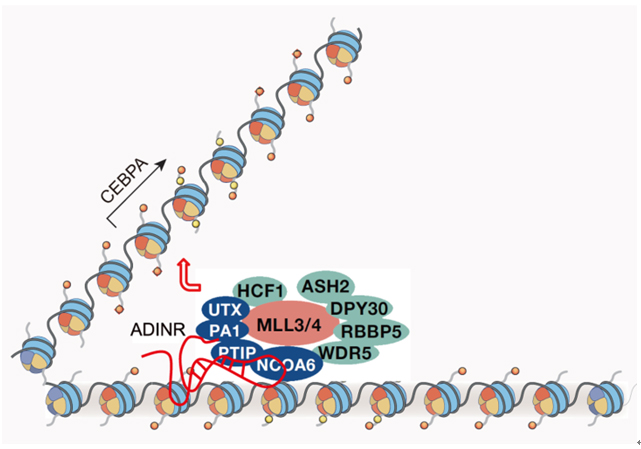
Figure: The graphic model of ADINR regulating adipogenesis.(© Stem Cell Reports) Contect
CHEN Runsheng
Institute of Biophysics, Chinese Academy of Sciences
Website: http://english.ibp.cas.cn/ibp_pi/CASMPI/201401/t20140115_116073.html
Email: chenrs@sun5.ibp.ac.cn |