A research article entitled “The zinc transporter ZIPT-7.1 regulates sperm activation in nematodes” is published online in PLoS Biology on June 7, 2018. This work identified a zinc transporter ZIPT-7.1 in nematode C. elegansand characterized the signal pathways by which ZIPT-7.1 functions to modulate sperm maturation. ZIPT-7.1 acts at the end of the SPE-8 pathway, triggering sperm activation and mediating the release of zinc from internal stores in the immature spermatids. The released zinc functions as a second messenger to promote the differentiation of the round, still spermatids into the mature, motile spermatozoa. This study has advanced the knowledge for the fields of zinc biology and sperm maturation.
Zinc is essential for all life and has well-established functions as a co-factor for numerous proteins. Zinc binding is necessary for the tertiary structure of many of these proteins, such as zinc finger transcription factors, and zinc binding to many enzymes is critical for enzymes’ catalytic activity. Although it has been suggested that the changes in zinc concentration in specific compartments might have second messenger effects, it has been difficult to demonstrate this type of signaling. The best-established setting is the extracellular release of zinc during synaptic transmission, which changes the concentration of zinc in the synaptic cleft. By contrast, examples of zinc signaling that control cell fate and development are lacking.
Sperm activation is a fascinating example of cell differentiation, in which immotile spermatids undergo a rapid and dramatic transition to become mature, motile sperm. Because the sperm nucleus is transcriptionally silent, this transition does not involve transcriptional changes. Although C. elegans is a leading model for studies of sperm activation, the mechanisms by which signaling pathways induce this transformation remain poorly characterized.
Previously, Dr. Long Miao’s group at the Institute of Biophysics found the microelement zinc as a novel physiological sperm activator depending on SPE-8 pathway to trigger sperm maturation (Liu et al., Development 2013). In this study, Drs.Yanmei Zhao and Long Miao together with their collaborators (Dr. Andrew Singson from Rutgers University, Dr. Ronald Ellis from Rowan University and Dr. Kerry Kornfeld from Washington University Medical School) propose a new model for sperm activation. The inactive ZIPT-7.1 islocalized to the membranous organelles of spermatids, which contain much higher levels of zinc than the cytoplasm. When activation is triggered, a signal transduced by the SPE-8 group of proteins opposes SPE-4 and SPE-6. As their function decreases, ZIPT-7.1 becomes active and transports zinc into the cytoplasm. The resulting increase in cytoplasmic zinc promotes the phenotypic changes that are characteristic of activation, including the acquisition of motility. Thus, the release of zinc from internal stores is a key part of the signal transduction process that mediates sperm activation. This work that a zinc signal controls sperm activation places zinc signaling in a specific biological context, in which changes in cell identity cannot be mediated by changes in gene expression. Furthermore, they have identified a zinc transporter that is central to this activation process. Their discoveries show that signal transduction using zinc can control how cells differentiate during development.
Dr. Yanmei Zhao from Long Miao’s lab (Institute of Biophysics of the Chinese Academy of Sciences), Dr. Chieh-Hsiang Tan (Washington University) and Dr. Amber Krauchunas (Rutgers University) are the co-first authors in this paper. Dr. Ronald Ellis (Rowan University) and Dr. Kerry Kornfeld (Washington University) are the corresponding authors. The Ph.D student Zhiheng Yuan from Miao lab also contributes to this work. This work was supported by the National Natural Science Foundation of China, the National Basic Research Program of China, National Science Foundation and National Institutes of Health of America. PloS Biology also published a commentary paper entitled “Zinc: A small molecule with a big impact on sperm function” by Dr. Diane Chu in the same issue of journal.
Article link:https://doi.org/10.1371/journal.pbio.2005069
Comment link:https://doi.org/10.1371/journal.pbio.2006204
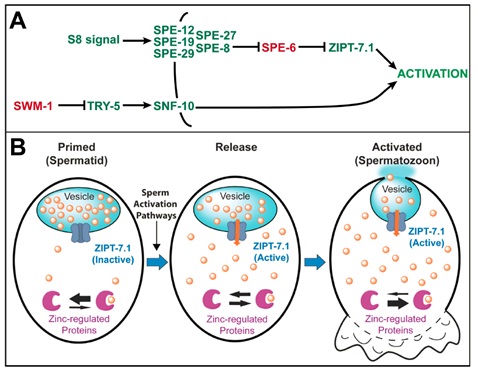
Model for ZIPT-7.1 function. (A) Model of the genetic function of zipt-7.1.
(B) Model of the biochemical function of ZIPT-7.1 at three times during sperm activation.
Contact: Yanmei Zhao
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
Phone: 86-10-64888478
Email: zhaoym(AT)ibp.ac.cn