Research reveals how cells modulate macropinocytic activity during the growth-to-development transition
Macropinocytosis mediates the non-selective bulk uptake of extracellular fluid, enabling cells to survey the environment and acquire nutrients. This process is observed in a variety of cell types and serves diverse functions. In the social amoeba Dictyostelium discoideum, macropinocytosis facilitates the uptake of nutrients, including amino acids and glucose, from the growth medium. In mammals, it plays an important role in nutrient acquisition, immune surveillance, synaptic activity regulation, and cell migration. Macropinocytosis is hijacked by pathogens, such as viruses and bacteria, to invade cells and is exploited by cancer cells to promote their growth and proliferation in the nutrient-poor tumor microenvironment by scavenging extracellular proteins and fatty acids.
Macropinocytic activity is regulated by diverse environmental factors. However, the mechanism of how cells adjust macropinocytic activity in response to environmental changes under different physiological or developmental contexts remains largely unknown. The laboratory strains of Dictyostelium provide a valuable model for studying macropinocytosis regulation. In nutrient-rich medium, these cells grow as individual amoeba and perform macropinocytosis at a high rate for nutrient acquisition. In contrast, upon starvation, up to 105-106 cells interact and aggregate to form multicellular structures. The early stage of this developmental program is under the guidance of oscillatory signals of cyclic adenosine 3',5'-monophosphate (cAMP). Periodic cAMP signals are generated, sensed, and relayed throughout the cell population, resulting in propagating waves of cAMP signals, which drive collective cell migration. Intriguingly, macropinocytosis is progressively downregulated during this transition from growth to development. Though this adaptive cellular behavior has been known for over 40 years, the underlying mechanism has not been elucidated.
In this study, we discovered that the transcription factors Hbx5 and MybG form a functional complex in the nucleus to maintain macropinocytic activity during the growth stage. In contrast, during starvation-induced multicellular development, the transcription factor complex undergoes nucleocytoplasmic shuttling in response to oscillatory cAMP signals, which leads to increased cytoplasmic retention of the complex and progressive downregulation of macropinocytosis. Therefore, by coupling macropinocytosis-related gene expression to the cAMP oscillation system that facilitates long-range cell-cell communication, the dynamic translocation of the Hbx5-MybG complex orchestrates a population-level adjustment of macropinocytic activity to adapt to changing environmental conditions.
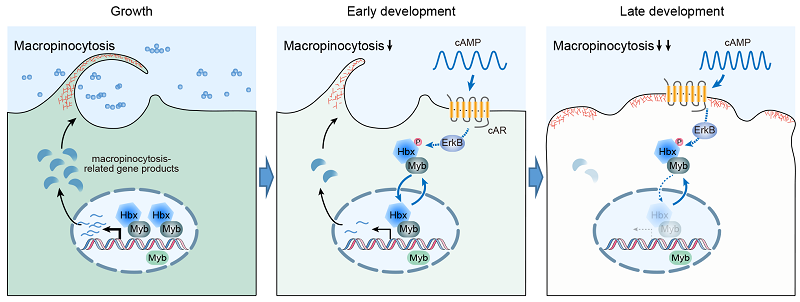
Figure. Hbx5-MybG transcription factor complex enables adaptive changes in macropinocytosis during the growth-to-development transition
Article link: https://doi.org/10.1016/j.devcel.2024.01.012
Contact: CAI Huaqing
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
Email: huaqingcai@ibp.ac.cn
(Reported by Prof. CAI Huaqing's group)

