Scientists Discover Transport and Analgesic Inhibitor Recognition Mechanisms of GlyT2
Glycine transporter 2 (GlyT2) serves as a critical modulator of inhibitory glycinergic neurotransmission, and its functional inhibition potentiates glycinergic signaling, making it a promising therapeutic strategy for neuropathic pain.
On November 24, a study in PNAS by Prof. ZHAO Yan's team at the Institute of Biophysics, Chinese Academy of Sciences, reported high-resolution cryo-EM structures of GlyT2 in three fundamental conformations, providing insights into its transport mechanisms and the molecular basis of analgesic compound recognition.
GlyT2 uniquely couples three Na+ ions and one Cl− ion for each cycle of glycine transport. The additional Na+ ion imparts enhanced substrate uptake efficiency that supports its specialized physiological role. The researchers identified a previously uncharacterized third sodium binding site (Na3), which provides the extra energetic drive required for transport and offers fundamental insights into Na+/Cl−-coupled substrate binding and conformational transitions.
The study also identified a unique allosteric binding pocket that accommodates lipid-based inhibitors, such as oleoyl-D-lysine, a derivative of the endogenous lipid N-arachidonyl glycine. Further analyses identified key characterization determining the inhibitory efficacy of this class of lipid inhibitors. These findings provide a structural rationale for designing optimized lipid-based GlyT2 analgesics.
In addition, the team resolved structures of GlyT2 bound to small-molecule inhibitors including ALX1393, opiranserin, and ORG25543. These structures reveal distinct binding modes for competitive and allosteric inhibitors and identify key residues that influence selectivity between GlyT1 and GlyT2.
Taken together, this study provides a comprehensive structural framework for understanding GlyT2-mediated glycine transport and inhibitor recognition. The discovery of the Na3 site, characterization of the lipid allosteric pocket, and mechanistic insights into diverse inhibitors open new avenues for the development of safer and more effective non-opioid pain treatments.
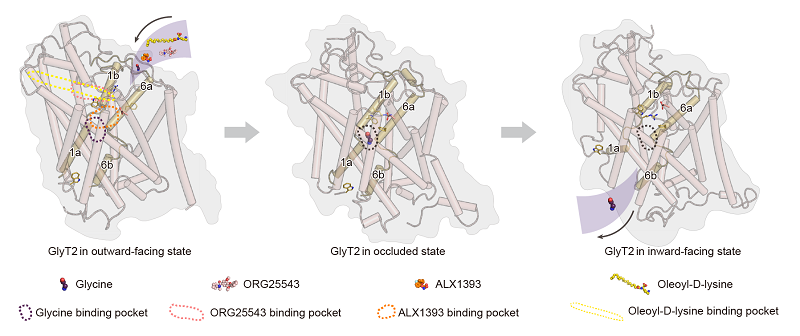
Figure: Binding modes of GlyT2 with substrate glycine and various analgesic inhibitors
(Image by ZHAO Yan's group)
Article link: https://doi.org/10.1073/pnas.2506722122
Contact: ZHAO Yan
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
E-mail: zhaoy@ibp.ac.cn
(Reported by Prof. ZHAO Yan's group)

