The endoplasmic reticulum displays high polarity with low protein aggregation in human cells
Protein homeostasis is fundamental to the maintenance of cellular physiological activities. Misfolding and aggregation of proteins are closely associated with the pathogenesis of neurodegenerative diseases and other protein conformational disorders. As a key physicochemical parameter of the cellular microenvironment, polarity, which is defined as dielectric constant, has been demonstrated in studies to regulate protein folding and aggregation in vitro. However, precise quantitative comparison of polarity across distinct subcellular compartments has not been reported, and the role of subcellular polarity in the regulation of proteostasis remains elusive.
On February 3, 2026, a study published in Communications Biology by researchers from the Institute of Biophysics of the Chinese Academy of Sciences, Westlake University, and the Dalian Institute of Chemical Physics of the Chinese Academy of Sciences revealed that the endoplasmic reticulum (ER) lumen exhibits a unique high-polarity, high-crowding environment, and subcellular environmental polarity is closely correlated with protein aggregation. The study uncovers a previously unrecognized regulatory mechanism of proteostasis mediated by subcellular physicochemical properties.
To measure subcellular polarity quantitatively, the researchers employed Halo proteins of distinct subcellular localization to achieve targeting the same polarity-sensitive fluorescent probe to different subcellular compartments. This strategy enabled quantitative determination and comparison of polarity in living cells. A crowding sensor was used to assess the crowding degree of different subcellular compartments, allowing the dissection of the relationship among subcellular polarity, crowding, and protein aggregation.
The researchers found that the ER lumen harbors a high-polarity and high-crowding environment and the feature is prevalent across various human cell lines. The high- polarity environment of the ER lumen remains stable during cellular aging and ER stress. The researchers further analyzed the correlation between polarity, crowding and protein aggregation, and found that the Huntingtin gene (Htt) exon1 with an expanded polyQ stretch (Htt-polyQ) has less tendency to form insoluble aggregates in the ER lumen (high polarity and more crowded) than in the cytosol (low polarity and less crowded). The study also observed that hypertonic treatments decrease polarity while increasing crowding. Under hypertonic treatment, the originally diffused Htt-polyQ in the cytosol further formed insoluble aggregates, indicating that polarity, rather than crowding, is correlated with protein aggregation. Protein homeostasis of the ER was previously attributed to its unique molecular chaperones, glycosylation modification, degradation pathways, high calcium environment and oxidative folding system. This study suggests that its intrinsic high-polarity environment may be important for resisting protein aggregation, and that the high-polarity environment of the ER lumen acts synergistically with its multiple regulatory mechanisms to construct a stable protein quality control network.
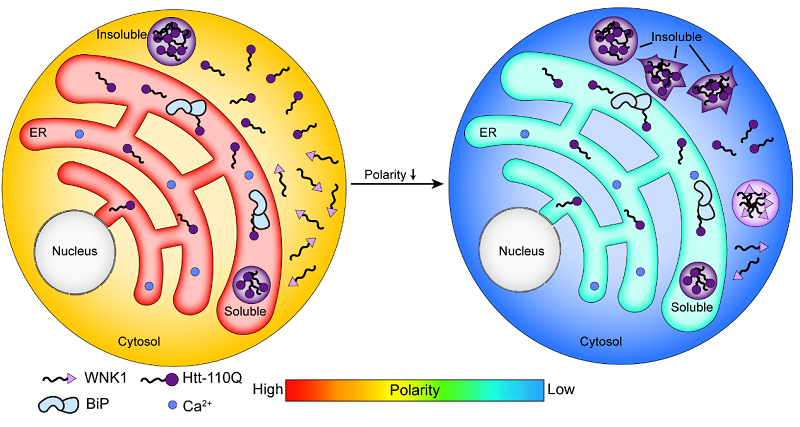
Figure: Schematic diagram of the model for polarity-regulated protein condensate and aggregate formation
(Image by WANG Lei's group)
Article link:
https://www.nature.com/articles/s42003-025-09491-w
Contact: WANG Lei
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
E-mail: wanglei@ibp.ac.cn
(Reported by Prof. WANG Lei's group)

