New Study Identifies GSNOR as a Key Driver of Age-Related Obesity
As people age, "middle-age weight gain" has become a widespread health problem and a major risk factor for metabolic diseases such as type 2 diabetes and cardiovascular disorders. However, the molecular mechanisms underlying this phenomenon remain incompletely understood.
On February 23, 2026, researchers from the Institute of Biophysics of the Chinese Academy of Sciences, in collaboration with Zhengzhou Central Hospital Affiliated to Zhengzhou University, published a study in Nature Communications, for the first time, identifying S-nitrosoglutathione reductase (GSNOR) as a novel age-related obesity gene.
The study reveals a previously unknown mechanism by which GSNOR drives adipose tissue "whitening" through regulation of protein S-nitrosation, thereby promoting age-related obesity, and highlights GSNOR as a potential therapeutic target.
The researchers first discovered that GSNOR expression is significantly elevated in the subcutaneous adipose tissue of middle-aged humans and mice compared with younger individuals.
Further investigations showed that deletion of GSNOR completely protects mice from age-related weight gain, whereas adipose-specific overexpression of GSNOR leads to an early-onset obesity phenotype. These findings establish GSNOR as a new gene associated with age-related obesity.
Overexpression of GSNOR in adipose tissue resulted in increased accumulation of white fat, accompanied by a marked reduction in thermogenic beige adipocytes responsible for energy expenditure. This shift toward subcutaneous adipose tissue "whitening" is a key driver of middle-age weight gain.
Using quantitative proteomic analysis of protein S-nitrosation, the researchers identified a critical target of GSNOR: the protein Beclin-1, a central regulator of autophagy.
GSNOR mediates the denitrosation of Beclin-1 at cysteine 351. This modification enhances the interaction between Beclin-1 and ATG14, thereby activating autophagy. Excessive autophagy accelerates mitochondrial clearance within cells, leading to the loss of beige adipocyte characteristics and promoting adipose tissue whitening, ultimately resulting in obesity.
Importantly, when GSNOR was knocked down in the adipose tissue of 14-month-old middle-aged mice, the level of adipose tissue beiging increased significantly and fat mass decreased. This finding demonstrates that targeting GSNOR offers a promising therapeutic window and strong translational potential.
Overall, this study is the first to identify GSNOR as a novel gene associated with age-related obesity and reveals a new mechanism in which redox-dependent protein S-nitrosation regulates middle-age weight gain. More importantly, it highlights GSNOR as a highly promising intervention target for the prevention and treatment of age-related obesity.
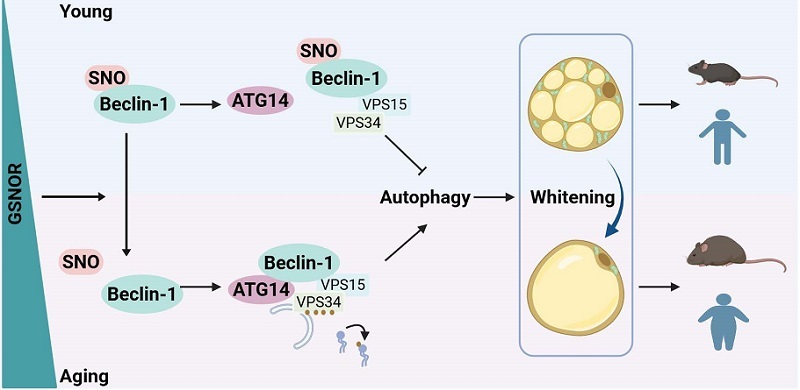
Figure. Schematic illustration of the molecular mechanism by which GSNOR promotes autophagy and induces adipose tissue whitening in age-related obesity.
(Image by CHEN Chang's group)
Article link: https://www.nature.com/articles/s41467-026-69793-3
Contact: CHEN Chang
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
E-mail: changchen@ibp.ac.cn
(Reported by Prof. CHEN Chang's group)

