A new ER-phagy mechanism: mechanosensory channels trigger autophagosome formation via Ca²⁺ transients
The endoplasmic reticulum (ER) is a highly dynamic intracellular membrane network composed of sheet-like cisternae and tubules. It is primarily responsible for protein synthesis and folding, and serves as the cell's main calcium (Ca²⁺) reservoir. When the ER experiences localized damage or stress, the cell activates "ER-phagy"-a selective autophagic process that acts as a cellular quality-control system. It maintains cellular homeostasis by forming autophagosomes to engulf and degrade the damaged ER fragments. However, the exact initiation signals, selective mechanisms, and membrane source of the autophagosome membranes in this process remain elusive.
On April 2, 2026, Prof. ZHANG Hong's group from the Institute of Biophysics, Chinese Academy of Sciences (CAS), published a paper in Molecular Cell titled "Mechanosensory channels mediate Ca²⁺ transients to trigger assembly of autophagosome initiation sites for degradation of ER subdomains." This study reveals, for the first time, the molecular mechanism by which mechanosensory channels mediate Ca²⁺ transients to trigger the degradation of ER fragments, offering new research directions for treating diseases related to ER dysfunction.
Using advanced super-resolution multi-SIM live-cell imaging, the researchers found that under stress conditions-such as prolonged starvation, cholesterol imbalance, or high Ca²⁺ insults-ER sheet subdomains containing high luminal Ca²⁺ expand and are then engulfed by autophagosomes for degradation. Surprisingly, electron microscopy and cryo-electron tomography revealed that the membranes of these autophagosomes originate directly from the remodeling of the ER. The study also found that this process relies on the collaboration of two key proteins (ER-phagy receptor FAM134B and lipidated LC3), while traditional autophagy proteins (ATG14 and ATG9) are not required.
Further, the researchers demonstrated that two mechanosensory channel proteins, PIEZO1 and TRPV1, are enriched on these high-Ca²⁺ ER sheets. They can sense changes in calcium concentration and trigger localized calcium release, inducing local Ca²⁺ transients. This signal drives liquid-liquid phase separation (LLPS) of the FIP200 initiation complex, ultimately triggering ER-phagy.
Imbalances in ER Ca²⁺ levels and dysfunctional ER-phagy are closely linked to the development of various diseases, including neurological disorders and cancer. By uncovering how stress induces the selective autophagic degradation of high-Ca²⁺ ER fragments, this study provides vital clues for understanding the pathology of diseases related to ER Ca²⁺ imbalance and identifies potential therapeutic targets.
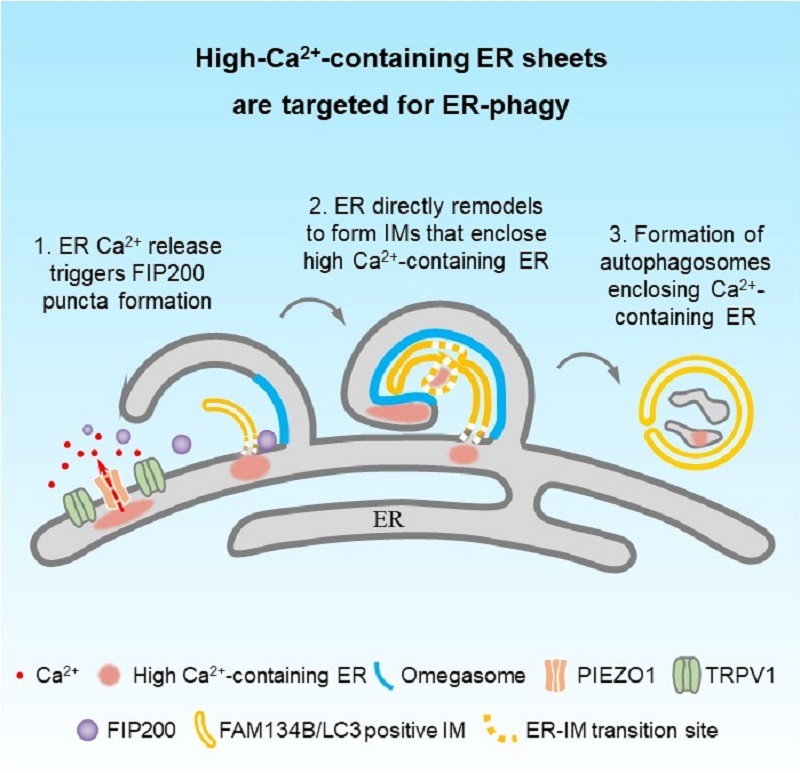
Figure Model of autophagosome formation containing high-Ca²⁺ ER
(Image by ZHANG Hong's group)
Article link: https://www.sciencedirect.com/science/article/pii/S1097276526001589
Contact: ZHANG Hong
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
E-mail: hongzhang@ibp.ac.cn
(Reported by Prof. ZHANG Hong's group)

