WANG Xiaoqun's group discovers Cenpj's regulatory functions to mammalian hypothalamus and food intake
The Science China-Life Sciences journal published a research paper titled "Loss of the centrosomal protein Cenpj leads to dysfunction of the hypothalamus and obesity in mice" on August 13, 2020. Jointly authored by the group of Professor WANG Xiaoqun with the Institute of Biophysics (IBP) under the Chinese Academy of Sciences (CAS) and the group of Professor WU Qian with Beijing Normal University, the research paper elaborates on the functions of centromere protein J (Cenpj) in regulating neurogenesis and food intake in the hypothalamus of mammalian species.
Hypothalamus, which grows from diencephalon, plays an important role in the regulation of appetite, weight and fat, glucose stability, and energy consumption arcuate nucleus (ARC) has been recognized as the feeding center of the brain. There are two typical ARC neurons. Neuropeptide-Y (NPY) and agouti-related peptide (AgRP) neurons are expected to facilitate food intake, while proopiomelanocortin (POMC) neurons exercise negative effects on food intake in accordance with surrounding nutrition and hormone signals. They work together to coordinate energy homeostasis signals with the aim of controlling food intake and steady development. Neurons of NPY/AgRP and POMC not only interact with each other but also send axonal projections to other areas of the brain, such as the paraventricular nucleus (PVN) of the hypothalamus. PVN neurons expressing melanocortin-4 receptors (MC4R) act to regulate energy balance. In the ARC, POMC neurons synthesize and secrete α-melanocyte-stimulating hormone (α-MSH) to activate PVN MC4R neurons and inhibit food intake. By contrast, AgRP/NPY neurons function as the inverse agonist of MC4R and promote food intake. These development changes connected with neuron projection in different groups lead to a profound effect on adults' body weight.
Cenpj is a centrosomal protein located at the centrosomes and the base of cilia. Previous research findings show that it plays an essential role in the regulation of neurogenesis and cerebral cortex development. Although centrosomal and cilium dysfunctions are among the causes of obesity and type-Ⅱ diabetes, the role that Cenpj plays in the regulation of body weight remains unclear.
Researchers employ Nkx2.1-Cre to produce conditionally knockout CenpjCKO mice by deleting their Cenpj genes. Four months later, those mice are inclined to eat more food, have decreased metabolism and become fat. The researchers find that Cenpj depletion will result in reduced proliferation and increased apoptosis at the embryonic stages, resulting in a dramatic decrease in the number of POMC neurons and electrophysiological dysfunction of NPY neurons in the ARCs. Meanwhile, the depletion of Cenpj also reduces neuronal projection from the ARC to the PVN, with decreased MC4R expression in PVN neurons. The study explains the biological mechanism in which the loss of Cenpj in Nkx2.1-expressing stem cells will exert influences upon the development of hypothalamus in two aspects. First, it is expected to reduce the number of POMC cells which inhibit food intake in the ARC, lessen MC4R α-MSH neurons in the PVN, and eventually block the inhibition of normal food intake. Second, NPY/AgRP neurons display high activity, possibly leading to a release of NPY/AgRP to neurons of the PVN (an agonist of MC4R) and stimulating food intake by acting on downstream receptors. Furthermore, the study gives an introduction to the roles Cenpj played in regulating hypothalamus development and body weight, and provides a foundation for further understanding of the pathological mechanisms of related diseases.
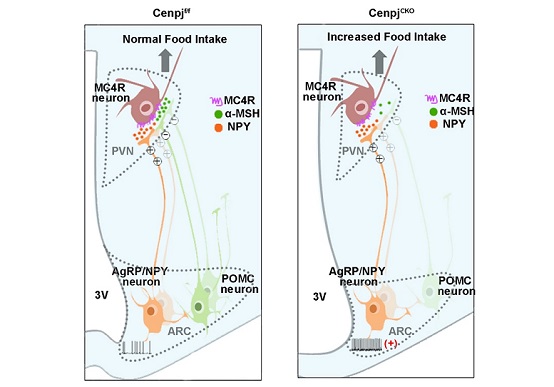
Figure 1: The loss of Cenpj results in dysfunction of hypothalamus and obesity in mice.
The work has been jointly accomplished by researchers from Beijing Normal University and the IBP under the CAS. Professor WU Qian with Beijing Normal University and Professor WANG Xiaoqun with the IBP under the CAS are correspondence authors of the research paper. Dr. DING Wenyu with Beijing Normal University and ZHANG Changjiang, a doctoral student at the IBP, are co-first authors of the published article. The study was funded by National Basic Research Program of China, National Natural Science Foundation of China, Strategic Priority Research Program of the CAS, and a research center affiliated to the Chinese Institute for Brain Research, Beijing.
The web link of this paper: https://engine.scichina.com/publisher/scp/journal/SCLS/doi/10.1007/s11427-020-1767-5?slug=fulltext
References:
Ding W, Wu Q, Sun L, Pan NC, Wang X. Cenpj regulates cilia disassembly and neurogenesis in the developing mouse cortex. Journal of Neuroscience. 2019 Mar 13; 39 (11): 1994-2010.
Contact: WANG Xiaoqun
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
Email: xiaoqunwang@ibp.ac.cn
(Reported by Dr. WANG Xiaoqun's group)

