On October 29, 2019, Dr. ZHANG Hong’s lab published a research article entitled “TGFβ-like DAF-7 acts as a systemic signal for autophagy regulation in C. elegans” in Journal of Cell Biology. This study found that the TGFβ-like molecule DAF-7 acts as a systemic factor that activates a canonical TGFβ signaling pathway in distant tissues to induce autophagy in C. elegans.
Autophagy is an evolutionarily conserved lysosome-mediated degradation process, involving the sequestration of a portion of cytosol in a double-membrane autophagosome and its delivery to lysosomes for degradation. In multicellular organisms, autophagy regulates diverse physiological functions such as stress resistance, cell fate determination and tissue remodelling. Studies of yeast and cultured cells have identified numerous factors that integrate various stressors with the autophagic machinery to modulate autophagy activity. In multicellular organisms, the stress response is coordinately controlled between different tissues/organs to ensure the maintenance of cellular homeostasis at an organismal level. Autophagy activity is systemically coordinated to ameliorate deleterious effects elicited by locally imposed stresses such as nutrient restriction, and also to maintain cell, tissue and organism homeostasis. For example, malignant tumors in fly eyes trigger autophagy in the tumor microenvironment and also induce autophagy in distant tissues, including the intestine, adipose tissue and muscle. Systemic intercellular signals that elicit the long-range autophagy response in distant cells/tissues/organs, however, remain elusive.
C. elegans is enclosed within a cuticle structure that serves as an impervious barrier between the animal and its environment. The cuticle is a multilayered extracellular matrix, composed predominantly of cross-linked collagens. The outer layer of the cuticle contains alternating parallel circumferential bands, known as annuli and annular furrows, which are comprised of two discrete interacting groups of collagens. Loss of function of annular furrow collagen genes, including dpy-2, -7 and -10, causes defects in cuticle integrity. Here we showed that during C. elegans development, loss of cuticle annular furrow collagens elicits autophagy in the hypodermis, intestine and muscle. The cilia of sensory neurons with cuticle-localized endings are essential for triggering this systemic response. The TGFβ-like molecule DAF-7, which is secreted in part from the ASI pair of ciliated neurons, acts as a systemic factor that activates a canonical TGFβ signaling pathway in distant tissues to induce autophagy. We also showed that AAK-2/AMPK and the STAT-like protein STA-2 act differentially in different tissues for autophagy activation. Our study reveals a circuit that senses and transduces the signal from the damaged cuticle to activate systemic autophagy during animal development.
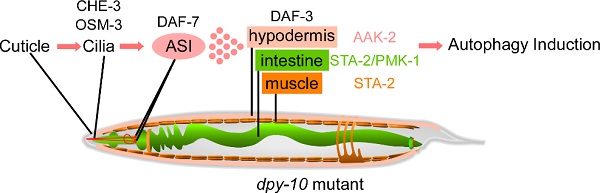
Figure. A model for the role of the dpy-10 family genes in autophagy regulation.
(Image by Dr. ZHANG Hong's group)
Dr. ZHANG Hong from the Institute of Biophysics is the corresponding author. Dr. ZHANG Yujie from Dr. ZHANG Hong’s lab is the first author of this paper. Dr. QI Linxiang from Dr. ZHANG Hong’s lab also contributes to this work. This work was supported by the Beijing Municipal Science and Technology Committee, the Strategic Priority Research Program, CAS, NSFC, National Chinese Ministry of Science and Technology, and the Key Research Program of Frontier Sciences, CAS.
Article link: http://jcb.rupress.org/content/early/2019/10/25/jcb.201907196
Contact: ZHANG Hong
Institute of Biophysics, Chinese Academy of Sciences
Beijing 100101, China
Phone: 86-10-64848238
Email: hongzhang@ibp.ac.cn
(Reported by Dr. ZHANG Hong's group)